GLP-2TZ
GLP-2TZ is a dual GIP and GLP-1 receptor agonist investigated for its unique dual-incretin mechanism of action in metabolic pathway research. This compound enables simultaneous study of two distinct receptor signaling cascades. For research purposes only.
Weekdays before 2PM EST
With Every Batch
Tested in USA



 ACH
Zelle
ACH
Zelle
Research Overview
GLP-2TZ is a first-in-class dual GIP and GLP-1 receptor agonist. It features a C-20 fatty di-acid moiety enabling extended albumin binding (half-life ~5 days). This compound has demonstrated remarkable efficacy in clinical trials and its dual mechanism represents a paradigm shift from single-incretin approaches in metabolic research.
Mechanism of Action
GLP-2TZ simultaneously activates two incretin receptors: GLP-1R — enhances glucose-dependent insulin secretion, suppresses glucagon, delays gastric emptying, and reduces appetite via hypothalamic signaling. GIPR — potentiates beta-cell insulin response, improves lipid metabolism, and may enhance adipose tissue insulin sensitivity. The GIP component is thought to provide additive metabolic benefits and may explain the superior efficacy observed compared to GLP-1-only approaches. The C-20 fatty acid enables albumin binding for once-weekly dosing.
Experimental Applications
Used in dual incretin receptor signaling studies, biased agonism research (GIP vs. GLP-1 pathway balance), beta-cell function and mass studies, appetite neuroscience, adipose tissue biology, hepatic lipid metabolism, cardiovascular protection models, and comparative pharmacology against single-agonist and triple-agonist incretin compounds.
Compound Information
Technical specifications
What is GLP-2TZ?
| Type | Dual GIP/GLP-1 Receptor Agonist |
| CAS Number | 2023788-19-2 |
| Molecular Weight | 4,813.45 g/mol |
| Amino Acids | 39 |
| Molecular Formula | C₂₂₅H₃₄₈N₄₈O₆₈ |
Stability Information
Loading structure…
Estimated plasma concentration over time
Compound Specifications
| Compound Type | Dual GIP/GLP-1 Receptor Agonist (acylated) |
| Purity | ≥ 99% |
| Appearance | White lyophilized powder |
| Storage | -20°C long-term; 2-8°C short-term |
| Testing Methods | HPLC, Mass Spectrometry |
| Packaging | Sealed sterile vial |
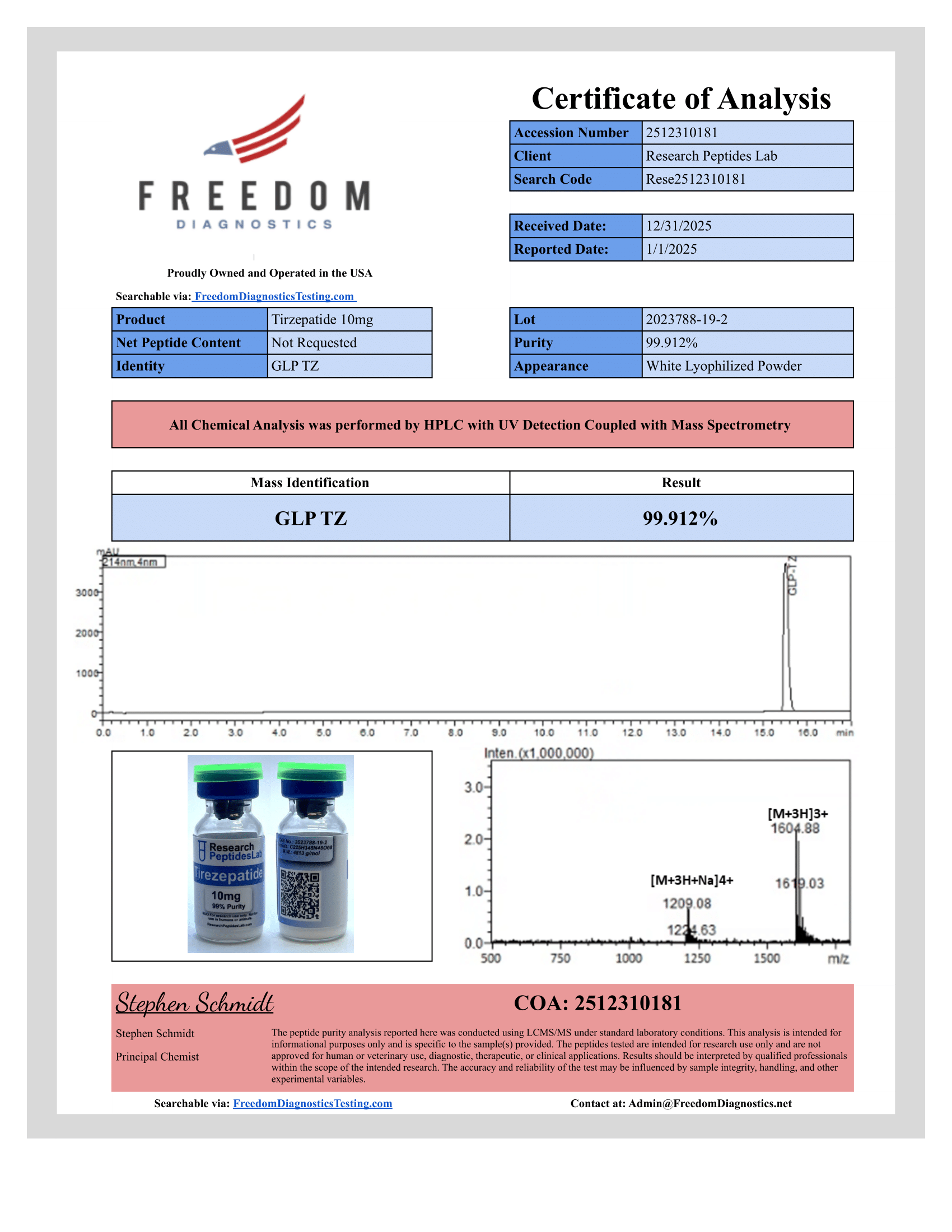
Certificate of Analysis
Lab Testing Notes
Each batch independently tested via HPLC (purity ?99%) and Mass Spectrometry (MW and identity confirmation). Acylation integrity verified. Full COA provided with every order.
Mechanism of Action
GLP-2TZ simultaneously activates two incretin receptors: GLP-1R — enhances glucose-dependent insulin secretion, suppresses glucagon, delays gastric emptying, and reduces appetite via hypothalamic signaling. GIPR — potentiates beta-cell insulin response, improves lipid metabolism, and may enhance adipose tissue insulin sensitivity. The GIP component is thought to provide additive metabolic benefits and may explain the superior efficacy observed compared to GLP-1-only approaches. The C-20 fatty acid enables albumin binding for once-weekly dosing.
Experimental Applications
Used in dual incretin receptor signaling studies, biased agonism research (GIP vs. GLP-1 pathway balance), beta-cell function and mass studies, appetite neuroscience, adipose tissue biology, hepatic lipid metabolism, cardiovascular protection models, and comparative pharmacology against single-agonist and triple-agonist incretin compounds.
Reconstitution Guidelines
Reconstitute with bacteriostatic water. Inject slowly along the vial wall. Gently swirl until dissolved — do not shake (acylated peptide). Recommended: 1–2 mL per vial. Store at 2–8°C. Use within 30 days.
How Does GLP-2TZ Compare?
GLP-1 & Metabolic Peptides
| Property | GLP-2TZ | GLP-1SG | GLP-3RT | AOD-9604 |
|---|---|---|---|---|
| Type | Dual GIP/GLP-1 Receptor Agonist | GLP-1 Receptor Agonist | Triple GIP/GLP-1/Glucagon Agonist | Modified hGH Fragment (176-191) |
| Molecular Weight | 4,813.45 | 4,113.58 | 4,731.28 | 1,815.08 |
| Amino Acids | 39 | 31 | 39 | 16 |
| CAS Number | 2023788-19-2 | 910463-68-2 | 2381089-83-2 | 221231-10-3 |
| Formula | C₂₂₅H₃₄₈N₄₈O₆₈ | C₁₈₇H₂₉₁N₄₅O₅₉ | C₂₂₁H₃₄₂N₄₆O₆₈ | C₇₈H₁₂₃N₂₃O₂₃S₂ |
| Current | View Product → | View Product → | View Product → |
Commonly Researched Together
Complementary compounds frequently paired with GLP-2TZ in published studies
Sources & References
Peer-reviewed research